Carbon Nanomaterial Formation at the Liquid-Liquid Interface
The liquid-liquid interface is a high-energy interface that can be stabilized using interfacially active molecules. The surfactants self-assemble at the oil-water interface, exposing their polar head group to water and apolar tail group to the oil phase, allowing the use of the liquid-liquid interface as template for two-dimensional assembly. Here we report the in-situ formation of 2D carbon nanomaterial at the liquid-liquid interface, starting from reactive carbon-rich surfactants based on hexaynes, that undergo spontaneous two-dimensional interfacial polymerization. Our laboratory had previously used carbon-rich reactive surfactants to fabricate carbon nanosheets at the air-water interface.[1] In the work presented here, we employ hexayne amphiphiles with a phosphonic acid head group that readily self-assemble at the oil-water interface and spontaneously react to form a cross-linked carbon-rich membrane. This membrane can be further crosslinked by UV irradiation, forming a thin 2D carbon nanomaterial at the interface. In contrast to molecular surfactants, the resulting nanomaterial present distinctive properties of a Pickering emulsion, such as buckling upon a change in the interfacial area. The chemical structure of the formed 2D nanomaterial is investigated in depth through various spectroscopic techniques. Furthermore, the transition from molecular surfactant stabilization to a colloidal stabilization of the interface can be followed by drop tensiometry as well as interfacial rheology and allows to gain fundamental insights into the interfacial stabilization process.
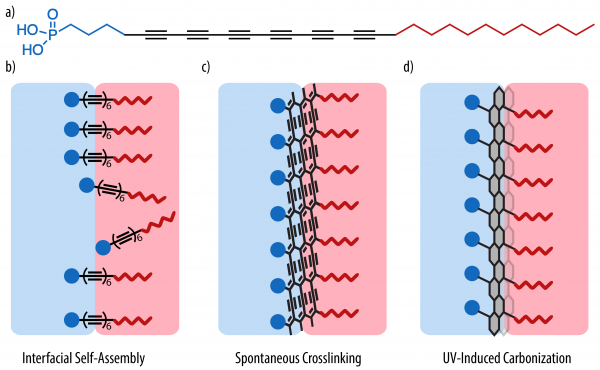
Figure 1. 2D carbon nanomaterial formation a) molecular structure of the surfactant b) self-assembly at the oil-water interface c) spontaneous crosslinking inducing the formation of a polydiacetylene-like membrane d) carbonization throught UV-irradiation into a carbon membrane
[1] Schrettl, S.; Frauenrath, H. et al, Nature Chem. 2014, 6, 468-476.