Ultrafast photoreduction of solvated iron compounds probed by VUV photoelectron spectroscopy
Transition metal complexes can exist in high-spin (HS) or low-spin (LS) configuration [1], whose equilibrium depends on oxidation state, nature of the ligands and coordination number. Light interaction triggers ultrafast electronic and structural dynamics such as bond cleavage, symmetry rearrangement, spin-crossover (SCO), and charge transfer [2]. These compounds can hardly be isolated, hence photochemistry of solutions is studied, where solvent plays a relevant role [3]. A good example is ferric trisoxalate [FeIII(ox)3]3- (ferrioxalate), that upon UV excitation experiences ligand dissociation because of forerunner processes yet to be clarified. The final photoproduct lacks of a ligand and the iron is reduced [4]. This implies a ligand-to-metal charge transfer (LMCT) process, a ligand-field strength reduction and a possible SCO. Recent X-ray studies at the iron K-edge concluded fragmentation within 3 ps [5]; other IR measurements demonstrated ligand bond breakage within the first ps [6]. Though, it is not yet clarified whether the metal reduction is a consequence of the cleavage or not, and how structural and electronic dynamics interplay at early times. We present a time-resolved study of photoexcited ferrioxalate aqueous solution by means of photoelectron spectroscopy (PES) [7-8], which is the only technique that provides the whole accessible electronic structure of the sample [9]. This allows us to simultaneously track electronic and structural dynamics of photoexcited solvated molecules. Upon 266 nm-wavelength excitation, we observe the Fe 3d photoelectron signal to shift towards the ferrous spectral position (see figure), i.e. metal photoreduction is prompt (<50 fs), which ascertains the LMCT as primary and driving mechanism. Furthermore, the transient photoelectron iron signal does not undergo peak narrowing whithin our probe range (5 ps), otherwise expected in case of HS to LS SCO [10]. These results, together with previous infrared [6] and X-ray studies [5], yield a complete picture of the photoinduced dynamics.
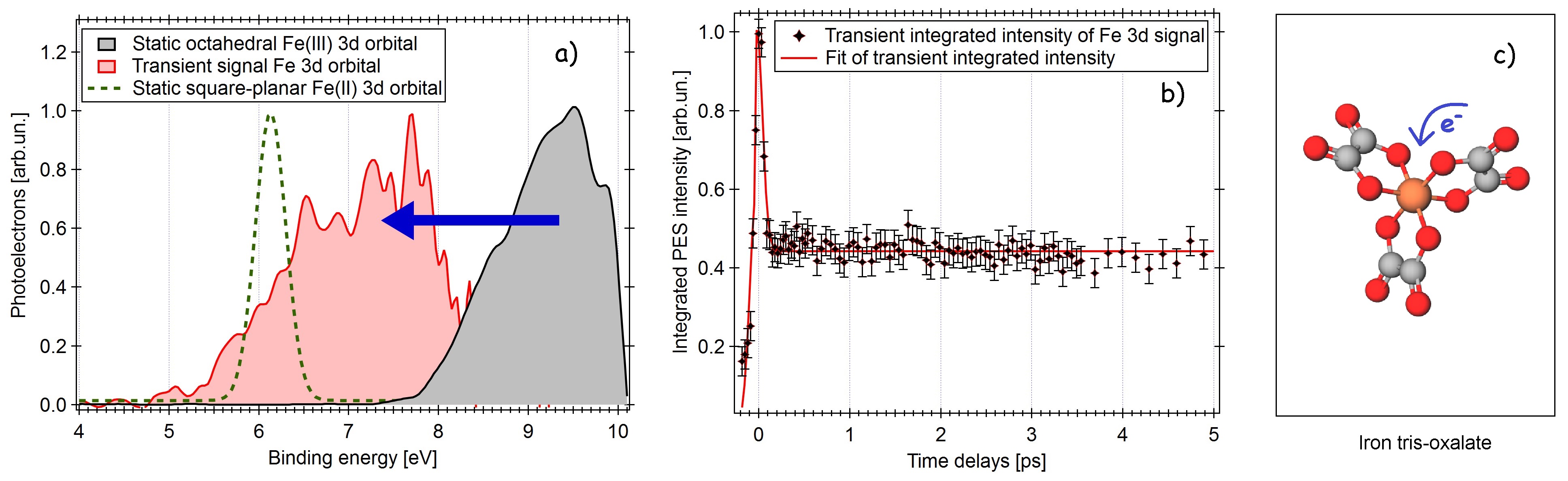
Figure: a) Static PES signal of Fe(III)oxalate iron 3d orbitals (black), and transient Fe(II) upon 266 nm excitation (red) shifting towards the final product Fe(II) spectral signature (dashed); b) the integrated PES Fe(II) transient intensity rises and remains constant; c) sketch of ferrioxalate with LMCT mechanism.
[1] J. Beattie, Advances in Inorganic Chemistry, 1988, 32, 1-53; [2] J. Ojeda et al., Phys. Chem. Chem. Phys., 2017, 19, 17052-17062; [3] T. Penfold et al., J. Phys. Chem. A, 2013, 117, 4591–4601; [4] Hatchard and Parker, Proc. Royal Society of London Series A, 1956, 235, 518-536; [5] Y. Ogi et al., Structural Dynamics, 2015, 2, 34901; [6] S. Straub et al., Phys. Chem. Chem. Phys., 2018, 20, 21390, [7] B. Winter and M. Faubel, Chem. Rev., 2006, 106, 1176−1211; [8] C. Arrell et al., Chimia, 2017, 71, 268-272; [9] J. Ojeda et al., Structural Dynamics, 2016, 3, 023602; [10] R. Seidel et al., J. Phys. Chem. B, 2011, 115, 11671-11677