Structure elucidation of low molecular weight compounds in spider venom
The venom of spiders is a mixture of proteins, peptides and low molecular weight compounds (< 1000 Da) such as nucleosides, amines and (acyl)polyamines. Several of these compounds were found to be promising lead compounds to treat cancer or neurodegenerative diseases like Alzheimer’s disease, epilepsy or stroke. [1] Yet, the knowledge about arachnotoxins is very limited asless than 1% of the venoms of the known spider species have been analysed up to date. Low available sample amounts, co-eluting isomers and use of trivial names in literature make the structure elucidation of low molecular weight compounds in spider venom demanding.
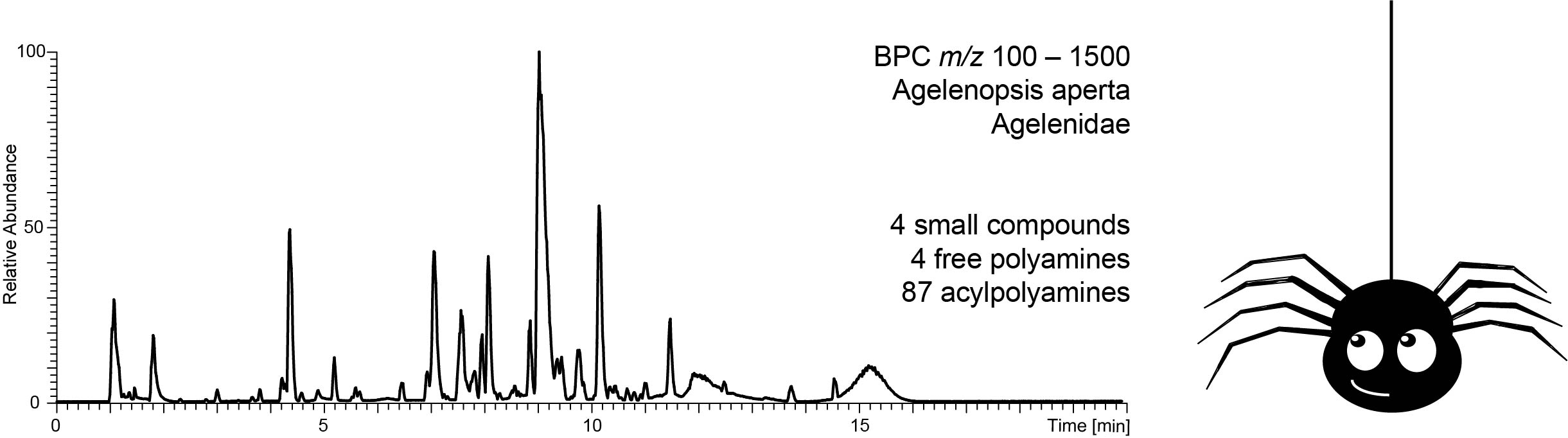
The spider venoms were analysed by UHPLC-HR-ESI-MS and MS/MS. Polyamines follow characteristic fragmentation rules; therefore MS/MS is ideal for their structure elucidation. [2] In addition, the number of acidic protons was determined in an on-line H/D exchange experiment (HDX). Moreover, venoMS, a freely accessible database for low molecular weight compounds in spider venoms, was created. The database provides fast access to related literature and acquired MS/MS spectra. Therefore, already known structures are identified easily and time can be used to focus on the structure elucidation of unknown lead compounds.
The fragmentation rules deduced from synthetic polyamine derivatives were used to develop and implement a fragment ion calculator allowing the structure elucidation of known and unknown acylpolyamines. Likely fragment ions are easily calculated by assembling the structure from simple building blocks.
The optimized analytical method in combination with venoMS enables a routinely annotation of known low molecular weight compounds and the structure elucidation of new acylpolyamines, even from limited sample amounts like spider venoms.
[1] Christian A. Olsen, Anders S. Kristensen, Kristian Strømgaard, Angew. Chem. Int. Ed. 2011, 50, 11296-11311.
[2] Manuel Tzouros, Serge Chesnov, Laurent Bigler, Stefan Bienz, Eur. J. Mass Spectrom. 2013, 19, 57-69.